AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
Back to Blog
Primary protein structure12/13/2023  Hydrophobic, aka non-polar, amino acids have saturated hydrocarbons as their side chains. Amino acids can, therefore, be divided into hydrophilic and hydrophobic groups. The most important property of amino acids that affects the folding and subsequently the function of the entire protein molecule is their known and predictable interaction with water. Essentially side chain makes the amino acid unique and can be thought of as its fingerprint. R symbolizes the variable side chain, which is the only chemical group that differs among all twenty amino acids. –COOH an acidic group (known as a carboxyl group).The α-carbon always has the following four groups attached to it: Each amino acid has a central carbon designated as the α-carbon. Properties of amino acids and their side chain differencesĪmino acids have the same base structure, which is important for proper chemical bond formation between adjoining molecules. Some of them make the cell and organism better adjusted to environmental pressures, a process you know as evolution. But not all mutations or protein modifications lead to disastrous consequences. Protein function can be jeopardized or lost completely if sequence is out of order. If you were to change one single arrangement of the chain, then that chain would have a completely different function. Each separate sequence has a unique order that conveys a very unique function. Conservation of specific protein sequences is so important that the cell has regulatory mechanisms in place to ensure that only perfect proteins are produced. A sequence of the amino acid chains provides diversity vital for meeting the demands of life. The polypeptide chain is composed of a sequence of amino acids dictated by the gene. How individual protein gets its identity lies in the ordered combination of amino acids, which determines all its characteristics.Īmino acids that are connected by a peptide bond are called a polypeptide chain. 
There are countless amino acids that exist in the world, but they are involved in other metabolic reactions but not protein synthesis. That does not mean there are only twenty amino acids. Only twenty amino acids are necessary and sufficient for generating thousands of proteins in a cell. They are synthesized from the template strand of DNA and contain unique and specific amino acid sequences in a linear form known as a primary structure. Proteins are linear polymers of amino acids connected by peptide bonds. The roles played by these molecules encompass anything from the transport of nutrients, catalyzing biochemical reactions to being structural components of cells or molecular motors. Proteins are the most important and versatile class of macromolecules in the cell. Every time a gene is transcribed, either over the lifetime of the cell or in any cell that has the same DNA, natural or recombinant, the proteins turn up alike and assume their pre-programmed function. This video shows the 4 levels of protein structure.Īdapted from RCSBProtein Data Bank under CC -BY licenceĪll that is needed to give a protein unique shape and therefore a unique function is “written” in a fragment of the DNA known as a gene. Quaternary structure describes the arrangement of the polypeptide chains in the multi subunit arrangement. Tertiary (3D) structure is a shape resulting from folding of secondary structures determined by interactions between side chains of amino acids. 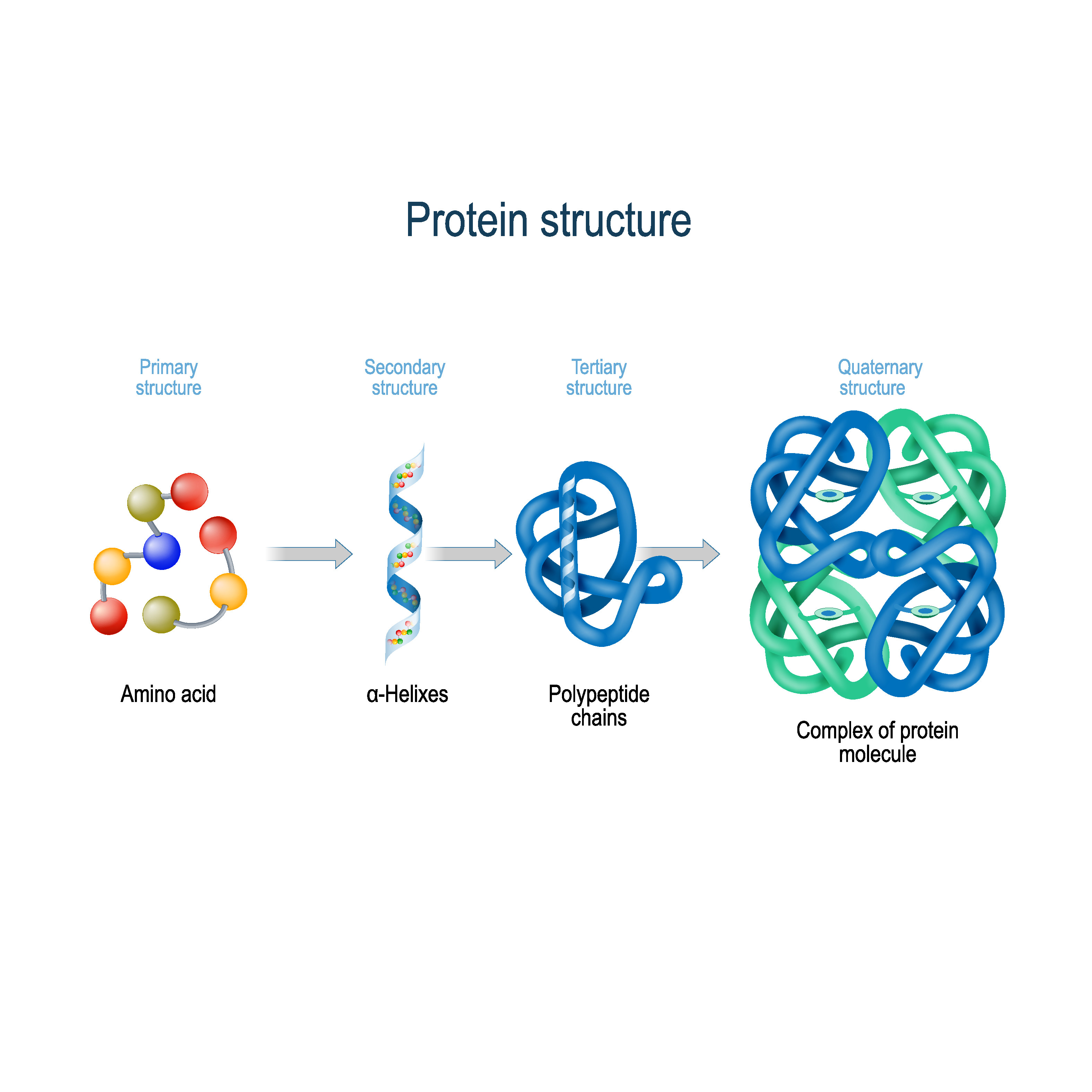
The secondary structure consists of local packing of polypeptide chain into α-helices and β-sheets due to hydrogen bonds between peptide bond – central carbon backbone. Primary structure refers to the linear sequence of the amino acids connected by the peptide bonds. Proteins have four levels of organization. Since the sequence of the amino acids is contingent on the genetic code, the shape of the protein is encoded in the DNA. The 3D conformation of the protein depends on the interactions between amino acids in the polypeptide chain. In short, the better fit between two molecules, the better it functions, the more bonds can be made, the faster the signal can pass, or the stronger two molecules connect (think adhesion molecules). The change of enzymatic pocket, due to mutation or modification of an amino acid residue changes the affinity and/or specificity of the enzyme. The best known example of the shape-function relationship is the “key and lock” theory of enzymatic function. The precise shape of the domain, resulting from the presence of non covalent bonds between residues in a polypeptide chain decides about the function. They are modular in nature and their interactions with other molecules in the cell rely on the presence of specific functional domains. Proteins are the building blocks of cell structures and motors of cellular activities. 3 Four levels of protein structure Protein structure and function 
0 Comments
Read More
Leave a Reply. |
 RSS Feed
RSS Feed
